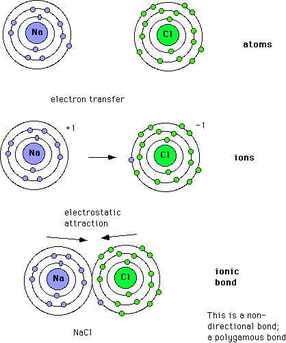
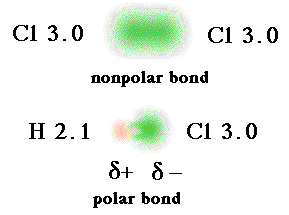
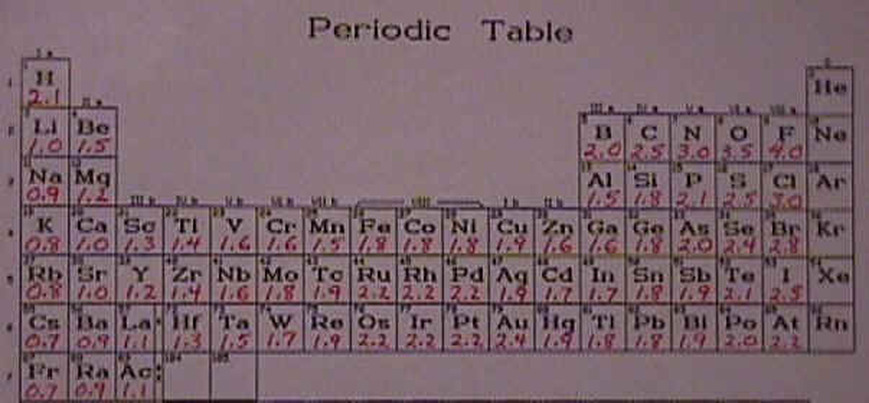
Electronegativity Values and Bond Designations
Bond Types
*Ionic Bonds can be formed when the difference in electronegativity between atoms is greater than 2.0.
Polar Covalent Bonds are formed when the difference in electronegativity between atoms is between 0.4 to 2.0.
Nonpolar Covalent Bonds are formed when the difference in electronegativity is less than 0.4.
|
* = Ionic Bonds aren't determined by their electronegativity, but by the transfer of an electron. They do, however, tend to happen between two atoms that share a greater electronegativity difference than 2.0.
Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. The Pauling scale is the most commonly used. Fluorine (the most electronegative element) is given a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7.
Electronegativity is determined by the amount of electrons in an atom's valence shell in comparison to a full amount of electrons possible in that atom's valence shell. So if the atom's valence shell has 1 electron and it can carry 8, it must have a low electronegativity, versus another atom which has 7 and can carry 8.
Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. The Pauling scale is the most commonly used. Fluorine (the most electronegative element) is given a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7.
Electronegativity is determined by the amount of electrons in an atom's valence shell in comparison to a full amount of electrons possible in that atom's valence shell. So if the atom's valence shell has 1 electron and it can carry 8, it must have a low electronegativity, versus another atom which has 7 and can carry 8.
The following image and information is from another website.
The electronegativity generally increases as you go from left to right across the periodic table. It decreases as you go down the periodic table.
I think you can see that the reason for this is going to depend on those same factors that we used to explain the trends in atomic size, ionization energy, and electron affinity.
First, the horizontal comparison. As you go across a period from left to right, the atoms of each element all have the same number of energy levels and the same number of shielding electrons. Thus the factor that predominates is the increased nuclear charge. When the nuclear charge increases, so will the attraction that the atom has for electrons in its outermost energy level and that means the electronegativity will increase.
Now what about the vertical comparison? As we established previously, when you go from one atom to another down a group, you are adding one more energy level of electrons for each period. The increased shielding nearly balances the increased nuclear charge and the predominant factor is the number of energy levels that are used by the electrons. So as you go from fluorine to chlorine to bromine and so on down the periodic table, the electrons are further away from the nucleus and better shielded from the nuclear charge and thus not as attracted to the nucleus. For that reason the electronegativity decreases as you go down the periodic table.
You might have noticed that there are some high values in the middle of the transition group. Those elements have fairly high electronegativities for metals. The reason for that ties in with the arrangement of electrons and the fact that the atoms are using d orbitals. The way that the d orbitals are shielded is different than the way that s and p orbitals are shielded so there is some variation in the transition metals that is not as easily explained as the general trend that I have just been talking about.
I think you can see that the reason for this is going to depend on those same factors that we used to explain the trends in atomic size, ionization energy, and electron affinity.
First, the horizontal comparison. As you go across a period from left to right, the atoms of each element all have the same number of energy levels and the same number of shielding electrons. Thus the factor that predominates is the increased nuclear charge. When the nuclear charge increases, so will the attraction that the atom has for electrons in its outermost energy level and that means the electronegativity will increase.
Now what about the vertical comparison? As we established previously, when you go from one atom to another down a group, you are adding one more energy level of electrons for each period. The increased shielding nearly balances the increased nuclear charge and the predominant factor is the number of energy levels that are used by the electrons. So as you go from fluorine to chlorine to bromine and so on down the periodic table, the electrons are further away from the nucleus and better shielded from the nuclear charge and thus not as attracted to the nucleus. For that reason the electronegativity decreases as you go down the periodic table.
You might have noticed that there are some high values in the middle of the transition group. Those elements have fairly high electronegativities for metals. The reason for that ties in with the arrangement of electrons and the fact that the atoms are using d orbitals. The way that the d orbitals are shielded is different than the way that s and p orbitals are shielded so there is some variation in the transition metals that is not as easily explained as the general trend that I have just been talking about.