Condensation and Hydrolysis
|
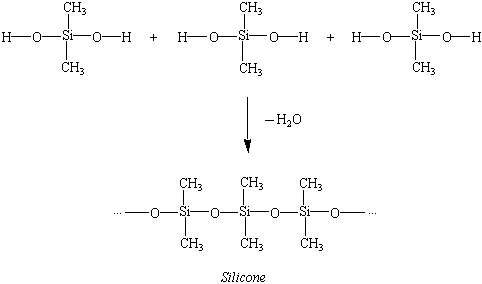
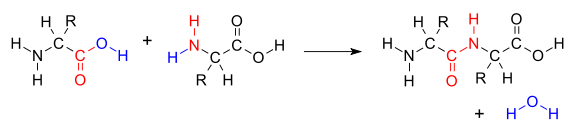
In the condensation example above we see three equal molecules with Oxygen (O) and Hydrogen (H) on both their left and right sides. What happens in condensation is that an O/H side of a molecule meets the O/H of another molecule and they covalently bond to release H2O (water), creating a compound . In this example, the remaining two Oxygen (O) atoms unite the three molecules to make Silicone with a glycosydic bond (linkage), and we lose two H2O (water) molecules. But we don't always have to have an oxygen (O) in between molecules; an example would be amino acids:
In the example above there are two groups of amino acids, one has an OH end while the other has an H end. When they join they create one H2O (water) molecule by covalently bonding, and the two amino acids join covalently as well.
|
In the hydrolysis example above we see a Sucrose compound [basically made up of a Glucose molecule and a Fructose molecule, joined by an Oxygen (O) atom in the center]. During hydrolysis, H2O (water) meets with the covalent bond, or a kind of linkage, of a compound and separates both parts, thus creates two distinct molecules. In this example we see that once the the H2O (water) molecule is integrated with Sucrose we end up with one Glucose molecule (which kept the O and one H from H2O) and one Frutose (which kept the other H from H2O).
|